- Blog
- Home
- Update Resource File Vb Net Tutorial Visual Basic
- Can Learner Drivers Carry Passengers Wa
- Conan The Destroyer Movie Torrent Download
- Primavera 6 Product Code Keygen Generator Crack
- Bcs Class 4 Drugs
- Cerere De Incetare Contract
- Merlin Vienna Soundfont Sf2 Free
- Kunguma Poove Konjum Purave Song Downloading
- Memories Of Murder 2003 Torrent
- Digital Electronics And Logic Design By J S Katre Pdf File
- Homer The Odyssey Robert Fagles Pdf To Word
- Mio Spirit 480 Crack
- Serial Dilution Sources Of Error In Measurement Definition
- Qurbani 1980 Free Mp3 Pk
- Hero Designer Crack Pipes
- Download Jamaican Gps Voice App
- Super Dvr Software For Windows 7 Download
- Big Money Deluxe Game Download
- Escribir Metro Cuadrado En Autocad Hay
- Dowlod Lagu Malaysia Dengan Dirinya
- Modelsim Pe Torrent Download
- Quicksurf 2007 Crack
- Daisy Powerline 790 Manual
- Cotabim Video 3gp
- Nfsu2 V1 2 No Cd Crack Please Insert Disc 2
- Greene King Retail Staff Handbook
- What Is Shaft Stiffness 6.0
- Free Golden Palominos Pure Rapidshare Programs In C
- Vampire Knight Destiny Episode 1 Sub Indo
BCS class 4 drugs for enhancement of oral bioavailability. For the same solid dispersion were prepared and analyzed for appropriate concentration of drug.
OPEN IN READ APP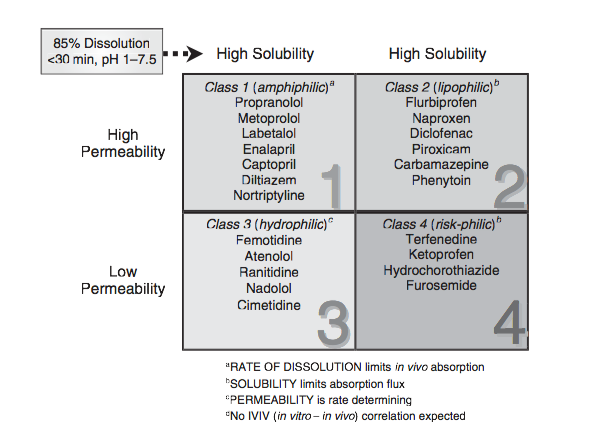
REVIEW
Journal of Controlled Release 2017 February 28, 248: 71-95
28088572
BCS class IV drugs (e.g., amphotericin B, furosemide, acetazolamide, ritonavir, paclitaxel) exhibit many characteristics that are problematic for effective oral and per oral delivery. Some of the problems associated include low aqueous solubility, poor permeability, erratic and poor absorption, inter and intra subject variability and significant positive food effect which leads to low and variable bioavailability. Also, most of the class IV drugs are substrate for P-glycoprotein (low permeability) and substrate for CYP3A4 (extensive pre systemic metabolism) which further potentiates the problem of poor therapeutic potential of these drugs. A decade back, extreme examples of class IV compounds were an exception rather than the rule, yet today many drug candidates under development pipeline fall into this category. Formulation and development of an efficacious delivery system for BCS class IV drugs are herculean tasks for any formulator. The inherent hurdles posed by these drugs hamper their translation to actual market. The importance of the formulation composition and design to successful drug development is especially illustrated by the BCS class IV case. To be clinically effective these drugs require the development of a proper delivery system for both oral and per oral delivery. Ideal oral dosage forms should produce both a reasonably high bioavailability and low inter and intra subject variability in absorption. Also, ideal systems for BCS class IV should produce a therapeutic concentration of the drug at reasonable dose volumes for intravenous administration. This article highlights the various techniques and upcoming strategies which can be employed for the development of highly notorious BCS class IV drugs. Some of the techniques employed are lipid based delivery systems, polymer based nanocarriers, crystal engineering (nanocrystals and co-crystals), liquisolid technology, self-emulsifying solid dispersions and miscellaneous techniques addressing the P-gp efflux problem. The review also focuses on the roadblocks in the clinical development of the aforementioned strategies such as problems in scale up, manufacturing under cGMP guidelines, appropriate quality control tests, validation of various processes and variable therein etc. It also brings to forefront the current lack of regulatory guidelines which poses difficulties during preclinical and clinical testing for submission of NDA and subsequent marketing. Today, the pharmaceutical industry has as its disposal a series of reliable and scalable formulation strategies for BCS Class IV drugs. However, due to lack of understanding of the basic physical chemistry behind these strategies formulation development is still driven by trial and error.
Discussion
You are not logged in. Sign Up or Log In to join the discussion.
Related Papers
Interplay of metabolism and transport in determining oral drug absorption and gut wall metabolism: a simulation assessment using the 'Advanced Dissolution, Absorption, Metabolism (ADAM)' model.Current Drug Metabolism 2010, 11 (9): 716-29
Spray freeze drying with polyvinylpyrrolidone and sodium caprate for improved dissolution and oral bioavailability of oleanolic acid, a BCS Class IV compound.International Journal of Pharmaceutics 2011 February 14, 404 (1): 148-58
Oral formulation strategies to improve solubility of poorly water-soluble drugs.Expert Opinion on Drug Delivery 2011, 8 (10): 1361-78
Formulation of poorly water-soluble drugs for oral administration: physicochemical and physiological issues and the lipid formulation classification system.European Journal of Pharmaceutical Sciences 2006, 29 (3): 278-87
Development and evaluation of novel solid nanodispersion system for oral delivery of poorly water-soluble drugs.Journal of Controlled Release 2013 July 10, 169 (1): 150-61
Self-emulsifying lipid formulation: an overview.Current Drug Delivery 2015, 12 (2): 166-76
Quantitative prediction of formulation-specific food effects and their population variability from in vitro data with the physiologically-based ADAM model: a case study using the BCS/BDDCS Class II drug nifedipine.European Journal of Pharmaceutical Sciences 2014 June 16, 57: 240-9
Self-nano-emulsifying drug delivery systems: an update of the biopharmaceutical aspects.Expert Opinion on Drug Delivery 2015, 12 (7): 1121-33
Biowaiver extension potential and IVIVC for BCS Class II drugs by formulation design: Case study for cyclosporine self-microemulsifying formulation.Archives of Pharmacal Research 2010, 33 (11): 1835-42
The Biopharmaceutics Classification System: subclasses for in vivo predictive dissolution (IPD) methodology and IVIVC.European Journal of Pharmaceutical Sciences 2014 June 16, 57: 152-63
Want full text access to this paper?
Use Read by QxMD to access full text via your institution or open access sources.
Read also provides personalized recommendations to keep you up to date in your field.
New to Read
3rd International Conference and Exhibition on Biowaivers, Biologics & Biosimilars
October 27-29, 2014 Hyderabad International Convention Centre, Hyderabad, India
Jithan Venkata Aukunuru
Keynote: J Bioanal Biomed
Abstract :
Bioavailability is the rate and extent (amount) of absorption of unchanged drug from its dosage form. It is one of the important parameter to achieve desired concentration of drug in systemic circulation for pharmacological response to be shown. A drug with poor bioavailability is one with poor aqueous solubility, slow dissolution rate in biological fluids, poor stability of dissolved drug at physiological pH, poor permeation through biomembrane, extensive presystemic metabol ism. From BCS candidates, class II and class IV drugs have solubility and permeability problems because of which their bioavailability is poor. Poorly water soluble drugs often require high doses in order to reach therapeutic plasma concentrations after oral administration. Low aqueous solubility is the major problem encountered with formulation development of new chemical entities. Any drug to be absorbed must be present in the form of an aqueous solution at the site of absorption. This presentation focuses on the various techniques used for the improvement of the Bioavailability of BCS class II and class IV drugs including size reduction, solubilising excipients, colloidal drug delivery systems, pH adjustment, solid dispersion, complexation, cosolvency, micellar solubilisation, hydrotropy etc.
Biography :
Jithan Venkata Aukunuru is presently a Professor and Principal at Mother Teresa College of Pharmacy (Affiliated to Osmania University), Hyderabad. Jithan is a recipient of several awards and medals in his entire academic career. His research interests include Novel Oral Delivery Technologies; Exploratory Pharmaceutics; IVIVC; Implants; Nanosuspensions; Microspheres; Proliposomes; Prodrugs; Colon Drug Delivery; Nanotechnology; Chronotherapeutics; Novel Transdermal Delivery Methods; Exploratory Pharmaceutics; Drug Metabolism; Pharmacology (Retinal and Liver Disorders); Solubility Enhancement. He was awarded Ph.D in 2002 from University of Nebraska Medical Sciences, USA, in Pharmaceutical Sciences. For his PhD, he worked on retinal delivery of small and macromolecules. He is a fellow of Association for Biotechnology & Pharmacy and an active member of APP, APTI, IPA and IPGA.
- Blog
- Home
- Update Resource File Vb Net Tutorial Visual Basic
- Can Learner Drivers Carry Passengers Wa
- Conan The Destroyer Movie Torrent Download
- Primavera 6 Product Code Keygen Generator Crack
- Bcs Class 4 Drugs
- Cerere De Incetare Contract
- Merlin Vienna Soundfont Sf2 Free
- Kunguma Poove Konjum Purave Song Downloading
- Memories Of Murder 2003 Torrent
- Digital Electronics And Logic Design By J S Katre Pdf File
- Homer The Odyssey Robert Fagles Pdf To Word
- Mio Spirit 480 Crack
- Serial Dilution Sources Of Error In Measurement Definition
- Qurbani 1980 Free Mp3 Pk
- Hero Designer Crack Pipes
- Download Jamaican Gps Voice App
- Super Dvr Software For Windows 7 Download
- Big Money Deluxe Game Download
- Escribir Metro Cuadrado En Autocad Hay
- Dowlod Lagu Malaysia Dengan Dirinya
- Modelsim Pe Torrent Download
- Quicksurf 2007 Crack
- Daisy Powerline 790 Manual
- Cotabim Video 3gp
- Nfsu2 V1 2 No Cd Crack Please Insert Disc 2
- Greene King Retail Staff Handbook
- What Is Shaft Stiffness 6.0
- Free Golden Palominos Pure Rapidshare Programs In C
- Vampire Knight Destiny Episode 1 Sub Indo
